Verkazia for the Treatment of Keratoconjunctivitis is now Publicly Reimbursed in Quebec
Friday, 05 March 2021 06:33.PM
Santen Canada Inc., a subsidiary of Santen Pharmaceutical Co., Ltd. (hereinafter, Santen), a global company focused exclusively on ophthalmology, today announced that Verkazia™ (cyclosporine topical ophthalmic emulsion 0.1% w/v) eye drops for the treatment of severe vernal keratoconjunctivitis (VKC) is now listed on the Quebec provincial formulary.
Verkazia™ was approved for the treatment of severe vernal keratoconjunctivitis in children from 4 years of age through adolescence by Health Canada in 2018.1
"Santen is pleased to see Verkazia reimbursed for patients living with severe VKC in Quebec," said Carol Stiff, Head of Santen Canada. "As a company dedicated to Ophthalmology, we aim to reduce the loss of social and economic opportunities for people around the world due to eye conditions and we hope this decision will help provide more access to this underserved population of Canadians."
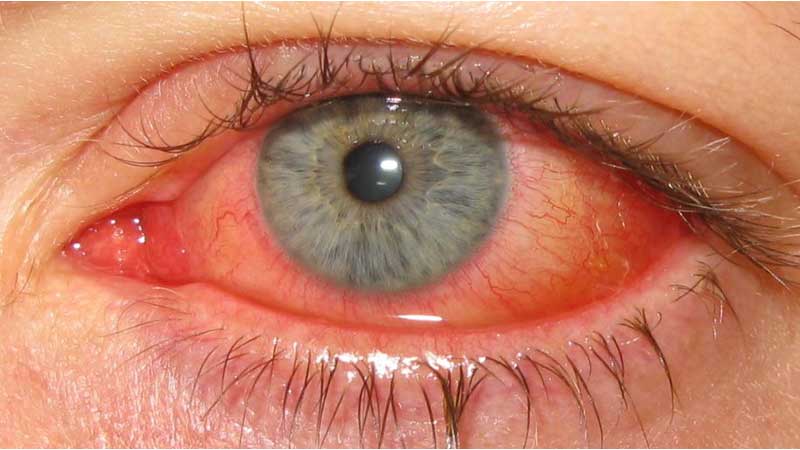
About Vernal Keratoconjunctivitis (VKC)
VKC is a rare and recurrent allergic eye condition, most common in children and adolescents, that causes severe inflammation of the surface of the eye. The main symptoms of VKC – light sensitivity, tearing, itching, and mucous discharge – can prevent those affected from participating in everyday activities.2,3,4 Approximately one-third of VKC cases are considered severe, and without adequate treatment may result in corneal ulcers and even vision loss.5
About Verkazia™
Verkazia™ cyclosporine 0.1% topical ophthalmic emulsion is indicated for treatment of severe vernal keratoconjunctivitis in children from 4 years of age through adolescence.1 The recommended treatment regimen approved by Health Canada is one drop of Verkazia 4 times a day (morning, noon, afternoon and evening) to be applied to each affected eye. The treatment should be discontinued if no improvement in signs and symptoms of vernal keratoconjunctivitis is observed. The treatment can be maintained at the recommended dose or decreased to one drop twice daily once adequate control of signs and symptoms is achieved. Treatment should be discontinued after signs and symptoms are resolved and reinitiated upon their recurrence.
SOURCE: Santen Pharmaceutical Co., Ltd.
- Related materials: