The JAMP Pharma Group Launches Prjamp Apremilast, A New Generic Alternative For The Treatment Of Plaque Psoriasis And Psoriatic Arthritis
Monday, 21 November 2022 02:20.PM
• The JAMP Pharma Group launches Apremilast, a specialty generic, offering a more affordable alternative to the reference product Otezla® (marketed by Amgen Canada Inc.).
• The JAMP Pharma Group, a leader in product launches1, expands its line of specialty products, contributing to its ambition to offer healthcare professionals and patients services adapted to their needs.
• The JAMP Pharma Group, established in Canada since 1988, contributes to the economic growth of the country and to a future where all Canadians can live a full and healthy life.
The JAMP Pharma Group, a Canadian pharmaceutical company based in the Montreal area, is proud to announce the launch of PrJAMP Apremilast, a generic alternative of Amgen's reference product Otezla®. PrJAMP Apremilast, available immediately, is indicated for the treatment of adult patients with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy. PrJAMP Apremilast, alone or in combination with methotrexate, is indicated for the treatment of active psoriatic arthritis in adult patients who have had an inadequate response, intolerance, or contraindication to a prior disease-modifying anti-rheumatic drug (DMARD). It is also indicated for adult patients with oral ulcers associated with Behçet's disease who are candidates for systemic therapy2.
"With more than 300 molecules in all segments of the pharmaceutical industry, JAMP Pharma Group is well positioned as a partner of choice in the Canadian market. The addition of Apremilast demonstrates our expertise and strong presence in the specialty drug segment", said Louis Pilon, President and CEO of JAMP Pharma Group. "Patients using this product will be supported by the JAMP Care™ Program, designed for patients and highly valued by healthcare providers".
It is estimated that nearly 1 million Canadians suffer from psoriasis, 90% of whom suffer from plaque psoriasis and 30% of whom also suffer from arthritis3. The arrival of this generic alternative, in a version of the same quality and more affordable, will allow better access for patients to this therapy, in addition to significant savings for drug plans.
JAMP Pharma stands out in offering this product in the form of a starter kit, just like the reference product. This allows PrJAMP-Apremilast to be taken gradually to the recommended dosage, which facilitates the work of physicians and pharmacists as well as the experience of patients.
"As dermatologists, we are happy to have Canadian companies like JAMP Pharma Group providing quality, lower cost options for our Psoriasis patients", says Dr Ron Vender, a Hamilton, Ontario Dermatologist and founder of Dermatrials Research Incorporated.
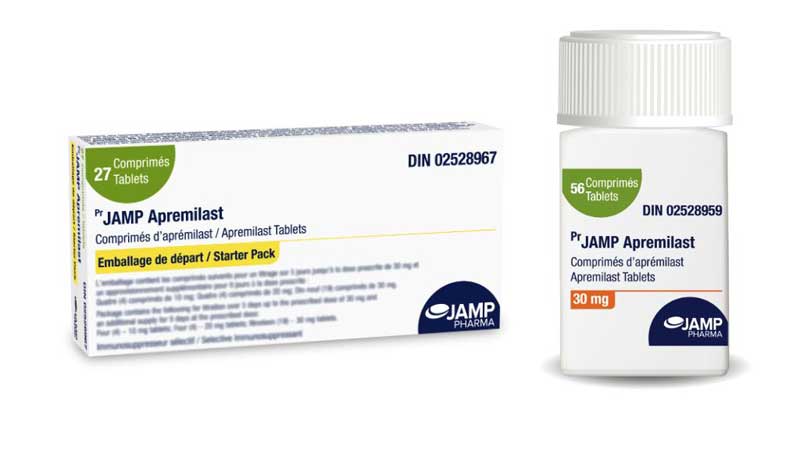
Strength and Sizes Available:
Starter Pack: 27-Count Starter Pack containing 10 mg, 20 mg and 30 mg tablets. Package contains two folded blister packs of 13 and 14 tablets per pack. Each of the blister packs identifies the tablets to be taken in the morning and evening according to the day of treatment.
Bottle of 30 mg: Bottle of 56 tablets with a safety cap.
SOURCE: JAMP Pharma